AZD 1390
AZD-1390
CAS: 2089288-03-7
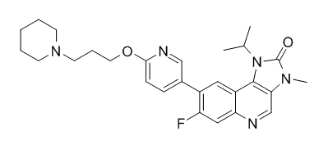
Molecular Formula: C27H32FN5O2
AZD 1390 - Names and Identifiers
AZD 1390 - Physico-chemical Properties
| Molecular Formula | C27H32FN5O2 |
| Molar Mass | 477.57 |
| Density | 1.224±0.06 g/cm3(Predicted) |
| Boling Point | 617.4±55.0 °C(Predicted) |
| Solubility | Soluble in DMSO |
| pKa | 9.22±0.10(Predicted) |
| Storage Condition | 2-8°C |
| Use | AZD1390 is an effective, highly selective, orally bioavailable, brain-permeable ATM inhibitor with an IC50 of 0.78 nM to ATM in cells. |
| In vitro study | AZD1390 inhibits the activity of DNA damage response dependent on ATM, and combined with radiation treatment can induce cell accumulation in the G2 cycle, causing micronucleus and apoptosis. Treatment with AZD1390 sensitizes glioma and lung cancer cell lines to radiation, and glioma cells carrying p53 mutants are more sensitive to radiation than glioma cells carrying wild-type p53. |
| In vivo study | AZD1390 had good oral bioavailability in preclinical studies in various animal models studied (66% in rats and 74% in dogs). In non-human primate PET studies, AZD1390 was able to effectively penetrate the blood-brain barrier. In an orthotopic xenograft brain tumor model, a combination of AZD1390 and radiotherapy for 2 or 4 days resulted in significant tumor regression and improved survival compared to radiotherapy alone. Combination of AZD1390 and ionizing radiation (whole brain or stereotactic radiotherapy) in in vivo, homologous or patient-derived glioma or orthotopic xenograft lung-brain tumor metastasis models, treatment with ionizing radiation alone may result in significant tumor regression and improved animal survival. AZD1390 has good physical, chemical, pharmacokinetic and pharmacodynamic properties, which is suitable for clinical drug development in the central nervous system. |
AZD 1390 - Preparation solution concentration reference
| 1mg | 5mg | 10mg | |
|---|---|---|---|
| 1 mM | 2.094 ml | 10.47 ml | 20.939 ml |
| 5 mM | 0.419 ml | 2.094 ml | 4.188 ml |
| 10 mM | 0.209 ml | 1.047 ml | 2.094 ml |
| 5 mM | 0.042 ml | 0.209 ml | 0.419 ml |
Last Update:2024-01-02 23:10:35
AZD 1390 - Introduction
AZD-1390 is a compound that belongs to the class of PARP (polymerase alpha transferase-related protein) inhibitors among antitumor drugs. It has the following properties:
-Appearance: AZD-1390 is a solid powder.
-Chemical structure: The chemical name is N-(8-fluoro-5-methyl-2-{[4-(methylsulfonyl)piperazin-1-yl]carbonyl}phenyl)-6-(1-methyl-1h-yrrolo [3,2-b]pyridin-3-yl)pyrazine-2-carboxamide.
-Solubility: AZD-1390 is soluble in some solvents, such as DMSO (dimethyl sulfoxide) and methanol.
-Stability: The AZD-1390 is stable at room temperature.
The main use of AZD-1390 is as an anti-tumor drug, by inhibiting the activity of PARP protein, blocking DNA repair mechanism, so as to improve the sensitivity of malignant tumor to chemotherapy drugs.
Regarding the production method, the detailed synthesis route of the AZD-1390 is not disclosed at present.
When using the AZD-1390, you need to pay attention to its safety information. Before use, read and follow the operating instructions on the product manual and relevant labels. According to the literature, some of the AZD-1390 safety information is as follows:
-toxicity: AZD-1390 in the preliminary experiment showed a certain toxicity, the use of the process should pay attention to safe operation.
-Storage: AZD-1390 should be stored in dry, dark and low temperature conditions, away from fire and oxidizing agents.
-Emergency measures: In case of contact with skin or eyes, rinse immediately with plenty of water for at least 15 minutes and seek medical help.
-Appearance: AZD-1390 is a solid powder.
-Chemical structure: The chemical name is N-(8-fluoro-5-methyl-2-{[4-(methylsulfonyl)piperazin-1-yl]carbonyl}phenyl)-6-(1-methyl-1h-yrrolo [3,2-b]pyridin-3-yl)pyrazine-2-carboxamide.
-Solubility: AZD-1390 is soluble in some solvents, such as DMSO (dimethyl sulfoxide) and methanol.
-Stability: The AZD-1390 is stable at room temperature.
The main use of AZD-1390 is as an anti-tumor drug, by inhibiting the activity of PARP protein, blocking DNA repair mechanism, so as to improve the sensitivity of malignant tumor to chemotherapy drugs.
Regarding the production method, the detailed synthesis route of the AZD-1390 is not disclosed at present.
When using the AZD-1390, you need to pay attention to its safety information. Before use, read and follow the operating instructions on the product manual and relevant labels. According to the literature, some of the AZD-1390 safety information is as follows:
-toxicity: AZD-1390 in the preliminary experiment showed a certain toxicity, the use of the process should pay attention to safe operation.
-Storage: AZD-1390 should be stored in dry, dark and low temperature conditions, away from fire and oxidizing agents.
-Emergency measures: In case of contact with skin or eyes, rinse immediately with plenty of water for at least 15 minutes and seek medical help.
Last Update:2024-04-09 21:49:45
Supplier List
Spot supply
Product Name: AZD1390 Visit Supplier Webpage Request for quotationCAS: 2089288-03-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: AZD-1390 Request for quotation
CAS: 2089288-03-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 2089288-03-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 7-Fluoro-1-isopropyl-3-methyl-8-(6-(3-(piperidin-1-yl)propoxy)pyridin-3-yl)-1,3-dihydro-2H-imidazo[4,5-c]quinolin-2-one Request for quotation
CAS: 2089288-03-7
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 2089288-03-7
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Spot supply
Product Name: AZD1390 Visit Supplier Webpage Request for quotationCAS: 2089288-03-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: AZD-1390 Request for quotation
CAS: 2089288-03-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 2089288-03-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 7-Fluoro-1-isopropyl-3-methyl-8-(6-(3-(piperidin-1-yl)propoxy)pyridin-3-yl)-1,3-dihydro-2H-imidazo[4,5-c]quinolin-2-one Request for quotation
CAS: 2089288-03-7
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 2089288-03-7
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
View History


